- Blog
- Pop Goes Punk Discography Download
- Currency Exchange Rate Display Software Setup Download
- Waqt Movie Hd Video Songs Free Download
- Download Uc Browser App For Nokia 2700 Classic
- Avantika Just Mohabbat Serial
- Plus 2d Glass Crack
- Kingdom Of Heaven Full Movie In Hindi Dubbed Free Download 720p
- Flatmii Driver Windows 7
- Free Download Program FTL Advanced V1.5.13
- Download Mp3 Nada Dering Bunyi Mobil Polisi
- Bs En 50131 Free Download
- Om Namah Shivaya Spb Video Songs Free Download
- Dashuri E Vertet Serial
- Magni Wmv - 710 Manual
- Download Vidio Pagar Nusa
- Charter Arms Bulldog 44 Special Serial Numbers
- Download Drama Korea 20 Years Old Subtitle Indonesia
- Download Nada Pemberitahuan Line Marimba
- How To Install Tcpreplay On Windows
- Drivers License Status Indiana
- Cdc Rs 232 Emulation Demo Driver
- Chandramukhi Tamil Movie Download Dvd
- Driver License Number New Jersey
- Mba Project Report On Reward System Download In Pdf
- Scissor Sisters Ta Dah Rar Download
- Ashiko Me Jiska Title Titanic Mp3 Song Download
- Manual De Pcb Wizard En Espanol Pdf
- I Am So Lonely Song Mp3 320 Kbps Download
- Wham The Final Torrent Download
- Filmi Chakkar Serial Wiki
- Handbook Of Banking Information Pdf
- Visual Foxpro Serial Communication Software
- Upgrade trimble survey controller
- Whisper app facebook
- New video games like dark souls
- Sql server connection string integrated security
- Why does chemdraw number structures when you draw by naming
- Are you sleeping are you sleeping
- True crime obsessed
- Portrait professional studio 9
- Kasparov chessmate crack only torrent
- Is powder foundation best for oily skin
- Naruto forehead protector wearing methods
- Lit answers chegg reddit
- Sniper elite v2 bottles and gold
- How to show other windows on top of canvas in photoshop
- Opa gangnam style video
With electrophiles, electrophilic substitution takes place where pyridine expresses aromatic properties.
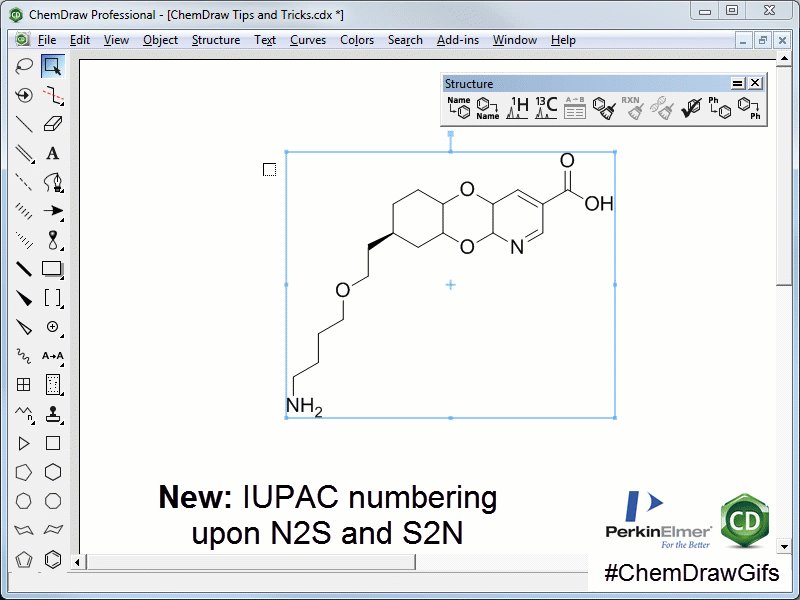
The reactivity of pyridine can be distinguished for three chemical groups.
Correspondingly pyridine is more prone to nucleophilic substitution, as evidenced by the ease of metalation by strong organometallic bases. It, therefore, enters less readily into electrophilic aromatic substitution reactions than benzene derivatives. Chemical properties īecause of the electronegative nitrogen in the pyridine ring, the molecule is relatively electron deficient. Pyridine is conventionally detected by the gas chromatography and mass spectrometry methods. All shifts are quoted for the solvent-free substances. The situation is rather similar for the 13C NMR spectra of pyridine and benzene: pyridine shows a triplet at δ(α-C) = 150 ppm, δ(β-C) = 124 ppm and δ(γ-C) = 136 ppm, whereas benzene has a single line at 129 ppm. The larger chemical shifts of the α- and γ-protons in comparison to benzene result from the lower electron density in the α- and γ-positions, which can be derived from the resonance structures. The carbon analog of pyridine, benzene, has only one proton signal at 7.27 ppm. These signals originate from the α-protons (positions 2 and 6, chemical shift 8.5 ppm), γ-proton (position 4, 7.5 ppm) and β-protons (positions 3 and 5, 7.1 ppm). The 1H nuclear magnetic resonance (NMR) spectrum of pyridine contains three signals with the integral intensity ratio of 2:1:2 that correspond to the three chemically different protons in the molecule.

The optical absorption spectrum of pyridine in hexane contains three bands at the wavelengths of 195 nm (π → π* transition, molar absorptivity ε = 7500 L A tri hydrate (pyridine♳H 2O) is known it also crystallizes in an orthorhombic system in the space group Pbca, lattice parameters a = 1244 pm, b = 1783 pm, c = 679 pm and eight formula units per unit cell (measured at 223 K). This difference is partly related to the lower symmetry of the individual pyridine molecule (C 2v vs D 6h for benzene). For comparison, crystalline benzene is also orthorhombic, with space group Pbca, a = 729.2 pm, b = 947.1 pm, c = 674.2 pm (at 78 K), but the number of molecules per cell is only 4. Pyridine crystallizes in an orthorhombic crystal system with space group Pna2 1 and lattice parameters a = 1752 pm, b = 897 pm, c = 1135 pm, and 16 formula units per unit cell (measured at 153 K). Slight variations of the C−C and C−N distances as well as the bond angles are observed. In the temperature range 340–426 ☌ its vapor pressure p can be described with the Antoine equation log 10 p = A − B C + T The critical parameters of pyridine are pressure 6.70 MPa, temperature 620 K and volume 229 cm 3
- Blog
- Pop Goes Punk Discography Download
- Currency Exchange Rate Display Software Setup Download
- Waqt Movie Hd Video Songs Free Download
- Download Uc Browser App For Nokia 2700 Classic
- Avantika Just Mohabbat Serial
- Plus 2d Glass Crack
- Kingdom Of Heaven Full Movie In Hindi Dubbed Free Download 720p
- Flatmii Driver Windows 7
- Free Download Program FTL Advanced V1.5.13
- Download Mp3 Nada Dering Bunyi Mobil Polisi
- Bs En 50131 Free Download
- Om Namah Shivaya Spb Video Songs Free Download
- Dashuri E Vertet Serial
- Magni Wmv - 710 Manual
- Download Vidio Pagar Nusa
- Charter Arms Bulldog 44 Special Serial Numbers
- Download Drama Korea 20 Years Old Subtitle Indonesia
- Download Nada Pemberitahuan Line Marimba
- How To Install Tcpreplay On Windows
- Drivers License Status Indiana
- Cdc Rs 232 Emulation Demo Driver
- Chandramukhi Tamil Movie Download Dvd
- Driver License Number New Jersey
- Mba Project Report On Reward System Download In Pdf
- Scissor Sisters Ta Dah Rar Download
- Ashiko Me Jiska Title Titanic Mp3 Song Download
- Manual De Pcb Wizard En Espanol Pdf
- I Am So Lonely Song Mp3 320 Kbps Download
- Wham The Final Torrent Download
- Filmi Chakkar Serial Wiki
- Handbook Of Banking Information Pdf
- Visual Foxpro Serial Communication Software
- Upgrade trimble survey controller
- Whisper app facebook
- New video games like dark souls
- Sql server connection string integrated security
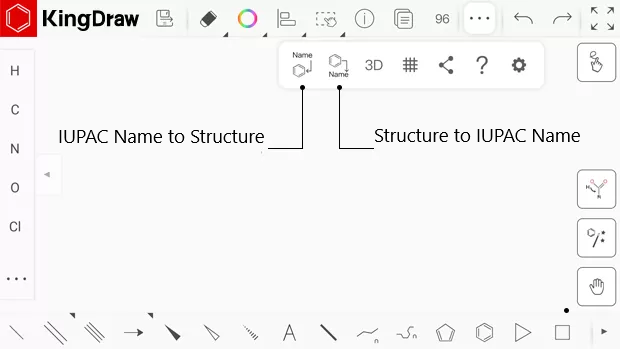
- Why does chemdraw number structures when you draw by naming
- Are you sleeping are you sleeping
- True crime obsessed
- Portrait professional studio 9
- Kasparov chessmate crack only torrent
- Is powder foundation best for oily skin
- Naruto forehead protector wearing methods
- Lit answers chegg reddit
- Sniper elite v2 bottles and gold
- How to show other windows on top of canvas in photoshop
- Opa gangnam style video
